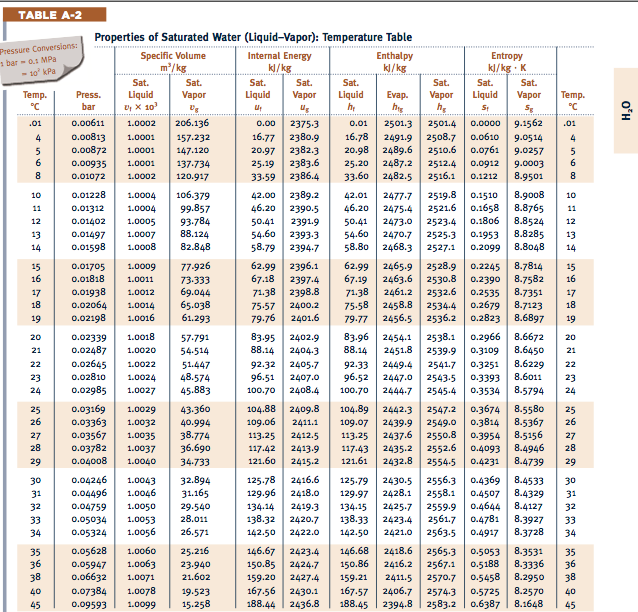
Although it isn't trivial in general, you can check how the formula simplifies for processes mentioned below. The general formula for work done by the gas is expressed as ∫p(V)dV if we consider pressure as the function of volume. These parameters in real gases differ from theoretical ones, but we already contain them in our thermodynamic processes calculator. Let our saturated water and steam calculator, based on NIST Steam Tables, be your reliable guide to steam and water thermodynamics. The Gibbs free energy equation is: small Delta G Delta H - (T cdot Delta S) G H (T S) where: G. R for gases with more complex molecules. With this entropy calculator, youll also be able to determine the change in Gibbs free energy using the results for entropy change that you get from other sections of the calculator.From known thermodynamic tables Kc at 500C for this reaction is 5.218E+002 which implies that at this temperature plenty of H 2 is made i.e. The base for the enthalpy in the table below is incompressible water at 0. ΔT, where Cv is molar heat capacity under constant volume. This page contains a steam table calculator and information on reaction rates.Internal energy change is proportional to temperature variation ΔT and type of gas with the following equation: ΔU = Cv Still, it is possible to find thermal energy changes ΔU, which are described by the first law of thermodynamics: ΔU = Q - W, where Q denotes heat absorbed, and W is work done by gas. 
It's quite tricky to estimate the precise value of internal energy. You can check by using CATT3 or similar thermodynamics table calculator A) Find enthalpy at T90C P0.1 MPa I B). Friend, Thermophysical Properties of Fluid Systems in NIST Chemistry WebBook, NIST Standard Reference Database Number 69, Eds. Internal energy U is the sum of all kinds of energy present in a system. Show your work and identify the phase as well. Enthalpy H 2551.013479 kJ/kg at 26.9 C and 0.0010 MPa.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
